S34.4: Foraging ecology of tropical seabirds
Lisa Taylor Ballance & Robert Lee Pitman
Ecology Program, National Marine Fisheries Service, Southwest Fisheries Science Center, 8604 La Jolla Shores Drive, La Jolla, California 92037 USA, fax 619 546 7003, e-mail lisa@caliban.ucsd.edu
Ballance, L.T. & Pitman, R.L. 1999. Foraging ecology of tropical seabirds. In: Adams, N.J. & Slotow, R.H. (eds) Proc. 22 Int. Ornithol. Congr., Durban: 2057-2071. Johannesburg: BirdLife South Africa.Tropical seabirds, species resident in waters ³ 23°C for at least one season of the year, forage in waters low in productivity relative to most non-tropical systems. Differential productivity of the world’s oceans has profound implications for seabirds; most tropical species must range widely and all are confined to forage essentially in two dimensions. Foraging strategies reflect these constraints. The single most important foraging strategy for tropical seabirds is to feed in multispecies flocks in association with subsurface predators, primarily tunas. In the tropical Pacific, these feeding opportunities support, at least in part, a majority of species, possibly a majority of individuals for some species, indirectly determine abundance and distribution, and provide a resource around which a complex community with a predictable structure is built. This degree of dependence has not been found in non-tropical seabirds. Other foraging strategies include solitary feeding and scavenging; few species use these exclusively, and those that do are rare. Nocturnal feeding has been observed regularly in only one species; at least two others purported to be nocturnal feeders obtain mesopelagic prey during the day by associating with subsurface predators. Further, because tunas are so important to seabird foraging success, and because tunas are diurnal feeders, nocturnal feeding is probably not significant in the tropics. Feeding in association with oceanographic features is important for non-tropical seabirds, but its significance in the tropics remains unknown. Important topics for future research include precise quantification of seabird dependence on tunas, on nocturnal feeding, and on oceanographic features as prey aggregating mechanisms, and investigation of scale-dependent patterns. The catastrophic decline of tropical seabirds following human colonisation of islands means that marine ornithologists are studying a remnant community that may not furnish unbiased data for proposing or testing ecological models. Thus, we suggest that our most important future research effort should focus on attempts to re-establish some of the former abundance and diversity of tropical seabird communities.
INTRODUCTION
The oceanic tropics are the major, central portion of the world ocean, comprising almost 50% of the total area of all open water on the Earth (Longhurst & Pauly 1987). Yet historically, our knowledge of tropical seabirds in general, and details of their foraging ecology in particular, have lagged far behind that of seabirds in temperate or polar oceans. At least three factors have contributed to this lack of knowledge.
First, logistics of research in the oceanic tropics are formidable. Tropical seabirds are thinly scattered over a huge area so that any effort to learn about them at sea requires months of ship time aboard expensive sea-going vessels. The same processes of scale that make tropical seabirds interesting to study, also make them difficult to study.
Second, as a consequence of logistic difficulties associated with oceanic tropical research, most of what is known about tropical seabird foraging ecology has been inferred from a few major food habit studies conducted on colonies (Ashmole & Ashmole 1967; Harrison et al. 1983; Diamond 1983; Imber et al. 1992). These studies, while insightful, are limited because they offer no direct information on how seabirds obtain the prey they bring back to colonies.
Third, tropical oceans are largely bordered by countries that do not have the resources to support research on marine ornithology. And most important fisheries, which inevitably affect seabirds in many ways, are found outside of the tropics. Consequently, the seabird literature is dominated by high latitude studies.
Thus a major goal of this paper will be to review what is known about the foraging ecology of tropical seabirds, and to highlight differences between tropical and non-tropical patterns. We offer definitions of the tropics and of tropical seabirds; we review what is generally known about their foods, feeding methods, and strategies for locating prey; we conclude with suggestions for future research.
DEFINITIONS
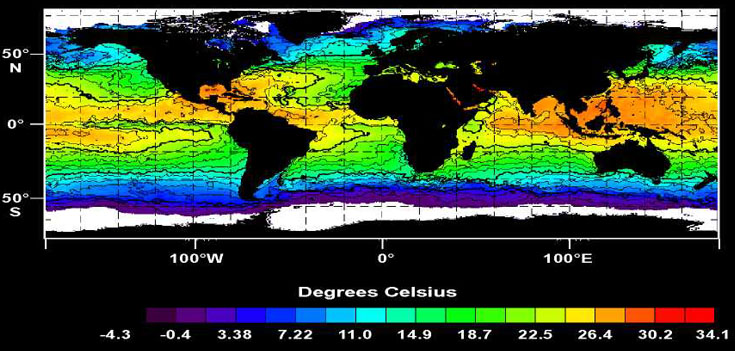
We define the tropics as ocean areas with sea surface temperatures ³ 23°C (Ashmole 1971; Fig. 1). This includes seasonally, as well as year-round, tropical waters. 'Non-tropical' will refer to areas with surface waters cooler than 23°C. Notably, some waters in tropical latitudes are not tropical habitat, including equatorial waters in the eastern Pacific, eastern Atlantic, and western Indian oceans (Fig. 1). Seasonal or year-round upwelling essentially makes these waters sub-tropical.
We define tropical seabirds as those that are resident in the tropics for at least one season of the year. Year-round residents include certain shearwaters and petrels (Procellariidae), certain storm-petrels (Oceanitidae), all tropicbirds (Phaethontidae) and frigatebirds (Fregatidae), all boobies (Sulidae), certain terns and all noddies (Laridae). Seasonal residents, species that breed in high latitudes and spend the non-breeding season in the tropics, include certain jaegers (Stercorariidae), certain phalaropes (Phalaropodidae), certain gadfly petrels Pterodroma spp., and certain storm-petrels. Although certain albatrosses (Black-footed, Laysan and Waved, Phoebastria nigripes, P. immutabilis, and P. irrorata, respectively) breed in colonies located on tropical margins, we do not include these as tropical species because it is likely that they are able to feed in non-tropical waters.
Throughout the paper we will compare tropical seabirds with 'non-tropical' seabirds. The latter includes species resident in temperate or polar waters in both northern and southern hemispheres, and trans-equatorial migrants (even though a few stragglers of the latter can often be found in the tropics year-round, e.g., Sooty and Pink-footed shearwaters, Puffinus griseus and P. creatopus; South Polar Skua, Catharacta maccormicki). Again, it is important to note that, by our definition, some seabirds inhabiting tropical latitudes are not tropical seabirds because they inhabit waters cooler than 23°C. Among the best examples are those species which breed and feed in the immediate vicinity of the Galápagos Islands (e.g., Galápagos Penguin Spheniscus mendiculus, Galápagos Cormorant Nannopterum harrisi).
In our review, we will take an at-sea perspective, focusing on what is known about the activities of tropical seabirds while they are at sea, attempting to locate and capture their prey. We discuss patterns primarily as they apply to the ocean seaward of the continental shelf, because this habitat comprises the vast majority of the tropical oceans. The data for this paper come from the published literature and from our own research in the eastern tropical Pacific and the western tropical Indian Ocean. Our eastern tropical Pacific data were collected aboard research vessels over a 17-year period (1976 - 1992), and they represent 1,781 days at sea distributed more or less evenly throughout all months and all years. Detailed methods can be found in Pitman (1986), Pitman & Ballance (1992), and Ballance et al. (1997). Our Indian Ocean data were collected during a four-month oceanic survey aboard a research vessel; methods are detailed in Ballance et al. (1996).
TROPICAL MARINE SYSTEMS AND IMPLICATIONS FOR SEABIRD FORAGING ECOLOGY
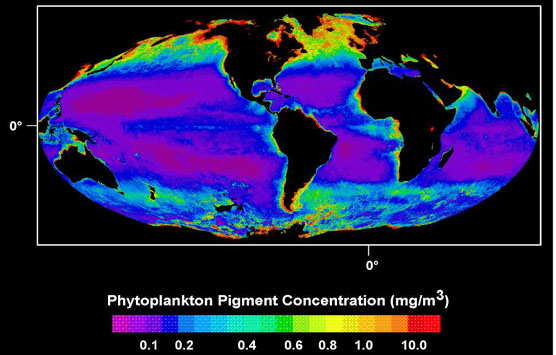
On a global scale, tropical marine systems are characterised by low productivity relative to non-tropical systems (the major central oceanic gyres excepted, Fig. 2). Simply put, in the tropics phytoplankton production is limited, and the total biomass at subsequent trophic levels is similarly affected. Marine ornithologists generally agree that for seabirds, low productivity equates with low prey abundance and patchy prey distribution (Ainley & Boekelheide 1983). Tropical seabirds are, therefore, under selective pressure to maintain foraging success in the face of diminished feeding opportunities relative to non-tropical species.
One parameter on which this selection might operate is proficiency of locomotion. For a homeothermic animal, cost of transport (energy expended per unit distance travelled) is lower for flight than for swimming (Peters 1983). So, tropical seabirds should be selected to travel through flight, and at low energetic cost. The few quantitative data available confirm that they are proficient flyers (e.g., Flint & Nagy1984; Ballance 1995).
Proficient flight carries a cost however, because the morphological adaptations for flight differ from those for swimming. For example, the avian wing can be designed to provide propulsion underwater or lift in the air, but in general the better suited it is for one, the less well suited it is for the other. This is clearly exemplified by deep-diving penguins which are flightless, and by aerially acrobatic species (e.g., frigatebirds, Sooty Tern, Sterna fuscata) which rarely or never land on the water (Ashmole 1963; Serventy et al. 1971; Harrison & Seki 1987).
Thus, with the development of proficient flight, tropical seabirds have largely lost their diving abilities and are confined to feed near or above the water surface. In contrast, non-tropical seabirds exhibit a wide spectrum of variation with respect to where they feed in the water column, ranging from obligate surface feeders (e.g. gadfly petrels, storm-petrels, terns, and phalaropes) to species that can dive to depths of hundreds of meters: penguins (Spheniscidae), auks (Alcidae), and cormorants (Phalacrocoracidae).
Thus, differential productivity of the world’s oceans has profound implications with respect to seabird foraging ecology: most tropical seabirds must range widely and all species forage essentially in two dimensions, while non-tropical seabirds include species that can exploit the ocean in three dimensions. The recognition of this link between productivity, flight proficiency, and adaptations for surface feeding is far from new (e.g., Ainley 1977; Brown 1980; Hulsman 1980; Ainley & Boekelheide 1983; Au & Pitman 1986). We reiterate it here because it provides an evolutionary basis for understanding the range of foraging strategies outlined below, and a context for comparison of tropical with non-tropical seabird communities.
FOOD AND PREY CAPTURE BEHAVIOUR OF TROPICAL SEABIRDSCompared with non-tropical species, the diets of tropical seabirds have been relatively little studied and nearly all research has been colony-based. Two major community-oriented studies are responsible for the bulk of knowledge (Ashmole and Ashmole 1967; Harrison et al. 1983; see also Diamond 1975a; 1975b; Schreiber and Hensley 1976; Diamond 1983; Imber et al. 1992). Of the fish and squid that comprise most of the diet for most tropical seabirds, flyingfish (Exocoetidae) and ommastrephid squid are almost always important and often the most common prey identified. But tropical seabirds consume a wide variety of prey; often diet diversity is much greater than that for non-tropical species (Harrison et al. 1983). Yet debate continues with respect to the significance, or even existence, of niche partitioning (e.g. Ashmole & Ashmole 1967; Diamond 1983; Harrison & Seki 1987).
Ashmole (1971) outlined the various ways in which seabirds capture prey in a classic paper. Prey capture methods of tropical seabirds reflect the fact that their adaptations for proficient flight constrain them to feed near, at, or above the water surface. Thus, feeding methods include plunge diving (boobies, tropicbirds), pursuit diving (the small black and white shearwaters), surface plunging (tropicbirds, terns), surface seizing (shearwaters, storm-petrels, gadfly petrels), kleptoparasitism (jaegers, frigatebirds), and aerial feeding (gadfly petrels, shearwaters, frigatebirds, Red-footed Booby, Sula sula; terns). This last technique, aerial feeding on marine prey, is unique to tropical seabirds and has presumably evolved in response to the abundance of volant prey (flyingfish and flying squid, see below).
HOW DO TROPICAL SEABIRDS LOCATE PREY?
The question of how seabirds locate prey in a seemingly featureless environment is far from completely answered. The discussion below summarises what is known about the strategies used by tropical seabirds. These strategies are not mutually exclusive; most species use more than one of the following methods. Nor are they restricted to tropical seabirds; many non-tropical species use them as well.
Associate with Subsurface Predators
Subsurface predators commonly drive prey to the surface because the air-water interface acts as a boundary beyond which most prey cannot escape. Under these circumstances, seabirds can access these same prey from the air; not surprisingly, association with subsurface predators is an important foraging strategy (review by Brown 1980).
Subsurface predators make prey available in at least three ways. First, simply by driving prey to the surface, predators prevent their escape to depth so that seabirds can access them (Ashmole & Ashmole 1967; Au & Pitman 1988). Presumably as a result of this pressure from below, some prey species in the tropics (the flyingfishes, Exocoetidae, and the flying squids, mostly Ommastrephidae) have evolved a flight escape response. While this has made these prey less vulnerable to predation from below, it has at the same time made them more vulnerable to predation from above, and as a group volant fish and squid comprise a substantial portion of the diet of many tropical seabird species (Ashmole & Ashmole 1967; Harrison et al. 1983). A second way in which subsurface predators make prey available to seabirds is by injuring or disorienting prey during pursuit, thus making them more vulnerable to capture. And third, while consuming prey, predators may leave scraps upon which seabirds can scavenge (Pitman & Ballance 1992).
Worldwide, subsurface predators include fish, cetaceans, pinnipeds, and other marine birds, but in the tropics the single most prevalent group is tunas (Ashmole & Ashmole 1967; Harrison and Seki 1987; Au & Pitman 1988). Tropical seabirds also associate with cetaceans, but these are of minor importance relative to tunas with respect to the number of species and number of individuals which they support (Au & Pitman 1986; 1988; Pitman & Ballance 1992; Ballance et al. 1997).
Seabird associations with subsurface predators have been recorded at virtually all latitudes (review by Brown 1980), and these feeding opportunities have been suggested as important for some non-tropical species (Obst & Hunt 1990; Grebmeir & Harrison 1992). But nowhere in the world is this strategy more significant than in the tropics.
The eastern and central tropical Pacific provide a strong example. Here, the single most important foraging strategy for seabirds as a group appears to be association with subsurface predators. Four lines of evidence support this conclusion. First, the vast majority of all seabird species in the eastern tropical Pacific have been recorded to feed in flocks, presumably in association with subsurface predators, at one time or another. This includes 90 different species from 27 genera (Ballance 1993): virtually all of the tropical residents plus some species that feed this way as they are migrating through the tropics. Although many of these use this strategy only occasionally, many do so often.
Second, for many species, the majority of feeding events seem to occur in association with subsurface predators. King (1970) reported that 70% of all birds sighted in the central Pacific were in flocks, presumably feeding with subsurface predators.
Third, there is a positive correlation between abundance of a seabird species and the strength of the subsurface predator association; species with highest abundances are those that regularly feed with subsurface predators. For example, in the eastern tropical Pacific, among the species recorded in association with subsurface predators most often and in greatest numbers is the Juan Fernandez Petrel (Pterodroma externa, King 1970; Au & Pitman 1986; Ballance 1993; Ballance et al. 1997), while one of the least common associates is the Red-tailed Tropicbird (Phaethon rubricauda, King 1970; Ballance 1993). In a ten-year survey (1974 - 1984, n = 4,333 survey hours, Pitman 1986), the number of Juan Fernandez Petrels recorded outnumbered Red-tailed Tropicbirds by two orders of magnitude: 11,410 petrels compared with 292 tropicbirds in the same sample. King’s data (1970) confirm this pattern for seabirds in the central Pacific. We predict that quantitative analysis will verify the positive correlation between absolute abundance and the strength of the subsurface predator association as well.
Finally, subsurface predators are significant enough as a resource that an entire community with a complex, yet predictable structure is built around them (Ballance et al. 1997). Seabirds associated with subsurface predators form flocks with characteristic species compositions. These 'flock types' exhibit largely disjunct distributions that reflect patterns in productivity. Species in different flock types show predictable gradients in body size and in cost-of-flight. In sum, this seabird community is predictably structured along a productivity gradient, ultimately as mediated through subsurface predators (see also Au & Pitman 1988).
Although tropical seabirds outside the Pacific have been much less studied, evidence indicates that the feeding opportunities that subsurface predators represent are of major importance in the Indian Ocean (Feare 1981), and the Caribbean (Shealer 1996).
Thus, although association with subsurface predators is important to seabirds worldwide, in the tropics it is of singular importance. Subsurface predators support, at least in part, the majority of species which feed in the tropics, and the majority of individuals for some species. These feeding opportunities indirectly determine distribution and abundance patterns, and provide the basis for a complex community with intricate interactions and a predictable structure. This degree of dependence has not been found in non-tropical seabirds.
Feed in Flocks
Flock feeding is another important strategy for tropical seabirds. It clearly is a result of feeding in association with subsurface predators, and this may have something to do with its significance. In fact, most feeding flocks in the oceanic tropics are associated with subsurface predators (Murphy & Ikehara 1955; Au & Pitman 1988). Although fish are often not directly detected by a human observer, the simple fact that tropical seabirds are confined to feed at the surface means that any persistent feeding flock must be associated with some force which prevents prey from escaping to depth.
However, there are at least three advantages to feeding in flocks in addition to those that come from association with subsurface predators. First, vulnerability of individual fish in a school may increase with an increase in the number of birds feeding in the flock (Götmark et al. 1986). Second, feeding flocks represent increased opportunities for kleptoparasitic species. Third, flocks are highly visual signals of the location of a prey patch (Hoffman et al. 1981). There is strong evidence that seabirds use these visual signals, in some cases distinguishing between searching and feeding seabirds, and between those feeding on a single prey item and those feeding on a clumped patch (Gould 1971; Hoffman et al. 1981). Depending upon the height of the flock and of an individual bird searching for prey, visual recruitment could occur from as far away as 30 km (Haney et al. 1992).
The importance of these behavioural signals cannot be underestimated, and this is true for seabirds in both the tropics, and in higher latitudes. Many studies report that feeding in flocks, often in a very few but very large aggregations, accounts for the majority of all individuals seen feeding (reviews by Hunt 1990; 1991b; see also Duffy 1983; Heinemann et al. 1989; 1990; Hunt et al. 1990; Veit and Hunt 1991; Pitman and Ballance 1992; Veit et al. 1993; Hunt et al. 1996). Whether this indicates that most feeding opportunities occur in patches, that most are best exploited by flocks, or simply that birds pay more attention to these visual signals than they do to patch quality (Obst 1985) is an issue that remains largely unexplored.
Feed at Night
Many fishes and invertebrates spend the daytime at depths of several hundred meters and migrate toward the surface after dark. During the night, surface densities of prey can be up to 1000 times greater than during the day (Brinton 1967). This migration is more significant in low than high latitudes, and in oceanic than neritic waters (review by Ashmole 1971). It seems likely then, that a good strategy for locating prey, particularly for pelagic birds in the tropics, would be simply to look for them at night.
Many studies of tropical seabirds infer nocturnal feeding from diet information. Specifically, the presence of vertically migrating species in seabird diets is taken as evidence that these seabirds must obtain their prey at night (review by Brown 1980; see also Imber et al. 1992; Harrison and Seki 1987). However, direct observation has confirmed nocturnal feeding in only three species that we know of, the Sooty Tern (Mörzer Bruyns and Boous 1965; Gould 1967), the Wedge-tailed Shearwater (Puffinus pacificus, Gould 1967), and the Swallow-tailed Gull (Creagrus furcatus, authors unpublished manuscript). Of these, only the latter feeds regularly at night (authors unpublished manuscript).
In contrast to inferences made from diet data, direct observation often confirms daytime feeding. At least two species, purported to be nocturnal feeders from information on diet, regularly obtain mesopelagic prey during the day, by associating with subsurface predators: Parkinson’s Petrel, Procellaria parkinsoni (Pitman & Ballance 1992) and the Dark-rumped Petrel, Pterodroma phaeopygia (authors unpublished manuscript). Further, because tunas are so important to foraging seabirds in the tropics, and because tunas are known to feed heavily during the day (Longhurst & Pauly 1987; although see Olson & Boggs 1986), we predict that nocturnal feeding is not important to most seabirds in the tropics.
Associate with Oceanographic Features
Physical features are known to increase prey abundance or availability in various ways (reviews by Hunt 1988; Schneider 1991). For example, physical gradients, including boundaries between currents, eddies, and water masses, can concentrate nutrients, and therefore enhance primary production. They can passively concentrate prey by carrying planktonic organisms through upwelling, downwelling, and convergence. And they can maintain property gradients (e.g., fronts) to which prey actively respond. Therefore, a good foraging strategy for a seabird is simply to locate these features. Not surprisingly, seabirds significantly and predictably associate with a wide spectrum of physical features, including fronts, sea ice, and topography (review by Schneider 1991).
It is important to note however, that virtually all of the supporting research linking feeding seabirds with oceanographic features has been conducted in non-tropical systems. Only a few tropical studies have identified this direct relationship (e.g., Pitman & Ballance 1990). More commonly, tropical studies report correlations between seabird distribution or abundance and water mass characteristics such as sea surface temperature and salinity without implicitly linking the association to foraging or feeding (Pocklington 1979; Ribic & Ainley 1997; Ribic et al. 1997). It is generally accepted that various oceanographic features and processes are important to the ecology of tropical tunas, although the precise relationships and effects are poorly understood (Blackburn 1965; Blackburn & Williams 1975). This can be taken as an indication that the link between physical features and many tropical seabirds is at least an indirect one, because of the strong dependence of these seabirds on tunas.
However, it may be that oceanographic features are of less importance to tropical seabirds. Physical features are prevalent in shelf systems where bathymetry interacts with currents to provide predictable locations where prey will be aggregated. But most continental shelf habitat occurs in non-tropical oceans (Longhurst & Pauly 1987). And, in tropical oceans there is less opportunity for physical forcing of planktonic organisms into dense aggregations, in part because there are not high densities to be advected.
In summary, the significance of oceanographic features to seabirds (or perhaps the scale at which they are manifested, see below) appears to differ between tropical and non-tropical oceans. But a definitive conclusion awaits further research.
Feed Alone
A few species seem to forage and feed almost exclusively alone. Tropicbirds are a good example. During a typical survey (1990, n = 97 survey days, Philbrick et al. 1991), we recorded 40 tropicbirds in 39 sightings, giving a mean of 1.02 individuals per tropicbird sighting. Among the most pelagic of species, they virtually never associate with subsurface predators or feed in flocks (King 1970; Ballance 1993), and they do not obviously feed at night or associate with oceanographic features. Tropicbirds are plunge divers and we assume that they search large areas for isolated prey items. We know little more about this foraging strategy.
Scavenge
Dead organisms floating at the surface are potential prey. They can range in size from tiny fishes to large squids and whales. But it is difficult to assess the extent to which seabirds rely on these items. Solitary scavengers sitting on the water are difficult to visually detect. And we know little about the abundance of dead prey at the surface, except that it appears to be rare.
Our impression from field research is that scavenging is limited to procellariiforms with heavy bills (Pterodroma spp., Pseudobulweria spp., Bulweria spp., some storm-petrels: Oceanodroma spp., Oceanites spp.), and is of relatively minor importance to all of these except possibly the Tahiti Petrel Pseudobulweria rostrata. However, on the few occasions when we have come across a dead whale floating at the surface (usually Sperm Whales, Physeter macrocephalus), there can be thousands of storm-petrels and up to five species feeding on the oil droplets leaching off the carcass.
Search Large Areas
All of the above strategies involve use of feeding opportunities that are patchy in both space and time, and often not predictable at small space or time scales. Under these circumstances, one of the most effective foraging strategies is simply to travel large distances in a continuous search for prey. Many tropical seabirds appear to do just this, ranging hundreds to thousands of km in a few days (Harrington 1977; Flint 1991).
This ability to search large areas is tightly linked to adaptations for flight proficiency, and, as noted above, we can expect that tropical seabirds should be so selected. For example, tropical seabirds should modify their flight behaviour to take advantage of the wind, and they appear to do just this (Harrington et al. 1972; Ainley & Boekelheide 1983; Schreiber & Chovan 1986; Ballance 1995; Spear & Ainley 1997a). Tropical seabirds should also exhibit morphological adaptations which allow for energy-efficient flight. Numerous researchers have suggested exactly this (Warham 1977; Ainley & Boekelheide 1983; Flint & Nagy 1984; Spear & Ainley 1997a). The precise links between flight behaviour, morphology, and energetic cost of flight have yet to be clarified. Clearly, these close relationships deserve more attention.
SYNTHESIS AND FUTURE DIRECTIONS
We believe that an important unifying theme in understanding the foraging ecology of seabirds as a group is their selective response to different productivity levels. The strategies for locating prey and methods for prey capture exhibited by tropical seabirds are largely a function of the fact that productivity on a global scale is low in the tropics relative to most non-tropical regions.
What are the prevailing questions and issues about tropical seabird foraging ecology that remain? We see that these fall into three major categories.
Basic Biology
We have yet to answer a number of basic questions about how tropical seabirds forage. For example, while it is widely accepted that subsurface predators are important, the exact degree of dependence remains largely unquantified. How important is this strategy to an individual bird or species? What proportion of feeding events occur in association with subsurface predators, and how much prey are obtained in this way?
Similarly, we still do not know the significance of night feeding. Many tropical seabirds obtain mesopelagic prey during the day, mostly through association with tunas. But whether these same seabirds also feed at night, and if so, how often, is not known. We suspect that in the near future, this issue will be resolved through the use of sophisticated remote devices designed to record behaviours of individual seabirds at sea (e.g., Weimerskirch & Wilson 1992).
Finally, while strong evidence from non-tropical research indicates that oceanographic features should be important to tropical seabirds, as of yet, this idea remains largely untested.
Theoretical Questions
Many theoretical issues remain unresolved with respect to seabird foraging ecology in general. For example, how effective are seabirds at locating prey patches and assessing their quality? How important is prey patch predictability? Can the visual signal of a feeding flock override the fact that there may be other prey patches nearby? What are the precise relationships between wind, wing morphology, flight behaviour, and the energetic cost of transport, within a species, between species, over a bird’s lifetime, over evolutionary time? More relevant here are theoretical questions that relate to differences between tropical and non-tropical seabirds. We give two examples below.
Scale
Scale-dependent patterns are patterns which vary with the resolution or range of measurement of a particular variable (Schneider 1994). The recognition of this effect has revolutionised the field of ecology in general (Wiens 1989; Schneider 1994), as well as many of its sub-disciplines, including seabird foraging ecology (e.g., Schneider & Piatt 1986; Briggs et al. 1987; Hunt & Schneider 1987; Hunt 1988; Heinemann et al. 1989; Cairns & Schneider 1990; Erikstad et al. 1990; Piatt 1990; Schneider et al. 1990; Hunt 1991; Hunt et al. 1992; Logerwell and Hargreaves 1996). Scale-dependence is relevant here because, as a group, tropical seabirds forage primarily over large spatial scales while non-tropical seabirds forage over a broad range of spatial scales, from small to large. This has several intriguing implications.
Sampling Methods and Scale
Survey methods for tropical seabirds have largely been adapted from those developed in high latitudes, where seabird densities are high and important processes occur at small spatial scales (though not exclusively). A typical method, developed in high latitudes but used throughout polar, temperate, and tropical seas, is the strip-transect method, typically used with a strip width of 300 m with data recorded continuously or in time period blocks (Tasker et al. 1984; Haney 1985; Gaston et al. 1987; Gould & Forsell 1989; Van Franeker 1990; Spear et al. 1992; Spear and Ainley 1997b). However, there is evidence that this method does not adequately sample the tropical seabird community or the important processes that occur there. For example, the feeding flocks that associate with subsurface predators, so important to tropical seabirds, are thinly scattered over large areas. So, in the eastern tropical Pacific in 1990 (97 survey days, Philbrick et al. 1991), we did not record a single flock using handheld binoculars and a standard strip width of 300 m. However, by using high powered mounted binoculars and a strip width of 8 km, we recorded 192 flocks during the same survey.
The reasons for this discrepancy are two-fold. First, many of the subsurface predator schools in the eastern tropical Pacific actively avoid large ships, taking their seabird flocks with them and outside of the 300 m sampling strip. But second, and we suspect more importantly, the spatial scale at which tropical seabirds forage may not be adequately sampled with a strip width of 300 m. Before we can correctly interpret patterns in tropical seabird foraging ecology, we will have to investigate how these differences in spatial scale affect sampling methods.
Energetics and Scale
A unifying theme in organismal ecology is that energy can be treated as a currency which is limited and which can be compared across species and ecosystems. All seabirds expend energy to locate prey. Tropical seabirds forage primarily at large spatial scales, while non-tropical seabirds forage at a range of spatial scales. With few exceptions (e.g., Obst et al. 1995) the relationships between energy expenditure and spatial scale of foraging have not received much attention.
Physiological ecologists often study physiological parameters in relative rather than absolute terms. An example of this approach can be found in the concept of metabolic scope, defined as the increase in metabolic rate between rest and maximal exertion (Schmidt-Nielsen 1984). This parameter is often expressed in terms of a ratio between the resting and maximal energy consumption, thus allowing for comparisons across behaviours and taxa.
This idea might be applied to energy and scale relationships for foraging seabirds, by using a relative measure, for example, the proportion of total available energy devoted to a particular behaviour. In this way, one might calculate the proportion of total available energy spent searching for prey, and compare this between tropical and non-tropical seabirds. While absolute energetic cost may differ, relative costs of foraging may approach a constant, regardless of species, or spatial scale. Clearly we need to know much more about these relationships; they may provide important insights into differences (or similarities) between tropical and non-tropical seabird communities.
Conservation
Steadman (1995, 1997) has chronicled a biodiversity crisis in the Pacific which began thousands of years ago, as humans began to colonise islands. The result was a progressive local extinction of seabirds on colony after colony. These extirpations were often long-term losses, not short-term departures of populations soon to be reestablished from elsewhere. For example, Huahine of the Society Islands historically supported more than fifteen breeding species of seabirds, today there are four; Ua Huka of the Marquesas historically supported more than twenty-two species, today there are four; Easter Island once supported more than thirty species of breeding seabirds, today there is one (Steadman 1995). This extirpation effect is greater in the tropics, where conditions are conducive to human survival, than in temperate or polar regions so that the total number of resident seabirds foraging in the tropical Pacific today may be as much as 100 to 1,000 times less than it was 3,000 years ago (Steadman 1997). Clearly, today’s global patterns of seabird distribution are not natural (Steadman 1995).
It may seem that conservation issues are far removed from the more academic topic of foraging ecology. However, the significance of this to marine ornithologists is that our attempts to understand seabird foraging ecology are based on studies that can not furnish unbiased data for proposing or testing ecological models. Even for those of us who study seabirds in what must surely be among the most unaltered habitat on the planet, the influence of anthropogenic effects is significant.
We suggest that the most important step we can take in the future is to try to re-establish some of the former abundance and diversity of tropical seabird communities by ridding colonies of introduced predators, and by re-introducing extirpated seabird species. It is only with a somewhat intact and functioning community, that we will be able to fully understand the answers to some of the questions we have posed here.
REFERENCES
Ainley, D.G. 1977. Feeding methods in seabirds: a comparison of polar and tropical nesting communities in the eastern Pacific ocean. In: Llano, G.A. (ed.). Adaptations within Antarctic ecosystems. Proceedings of the Third SCAR Symposium on Antarctic Biology. Washington, D.C; Smithsonian Institution:669-685.
Ainley, D.G. & Boekelheide, R.H. 1983. An ecological comparison of oceanic seabird communities in the south Pacific Ocean. Studies in Avian Biology 8:2-23.
Ashmole, N.P. 1963. The regulation of numbers of tropical oceanic birds. Ibis 103:458-473.
Ashmole, N.P. & Ashmole, M.J. 1967. Comparative feeding ecology of sea birds of a tropical oceanic island. Peabody Museum of Natural History, Yale University, Bulletin 24. Peabody Museum of Natural history, New Haven, Connecticut, U.S.A.
Ashmole, N.P. 1971. Sea bird ecology and the marine environment. In: Farner, D.S. and King, J.R. (eds.). Avian biology. Volume I. New York; Academic Press:223-286.
Au, D.W.K. & Pitman, R.L. 1986. Seabird interactions with dolphins and tuna in the eastern tropical pacific. Condor 88:304-317.
Au. D.W.K. & Pitman, R.L. 1988. Seabird relationships with tropical tunas and dolphins. In: Burger, J. (ed.). Seabirds and other marine vertebrates. Competition, predation, and other interactions. New York; Columbia University Press:174-209.
Ballance, L.T. 1993. Community ecology and flight energetics in tropical seabirds of the eastern Pacific: energetic correlates of guild structure. Ph.D. Thesis. University of California, Los Angeles, U.S.A.
Ballance, L.T. 1995. Flight energetics of free-ranging Red-footed Boobies Sula sula. Physiological Zoology 68:887-914.
Ballance, L.T., Pitman, R.L., Reilly, S.B. & Force, M.P. 1996. Report of a cetacean, seabird marine turtle and flyingfish survey of the western tropical Indian Ocean aboard the research vessel Malcolm Baldrige March 21 - July 26, 1995. NOAA Technical Memorandum NMFS. NOAA-TM-NMFS-SWFSC-224. Southwest Fisheries Science Center, 8604 La Jolla Shores Drive, La Jolla, CA 92037.
Ballance, L.T., Pitman, R.L. & Reilly, S.B. 1997. Seabird community structure along a productivity gradient: importance of competition and energetic constraint. Ecology 78:1502-1518.
Blackburn, M. 1965. Oceanography and the ecology of tunas. Oceanography and Marine Biology Annual Reviews 3:299-322.
Blackburn, M. & Williams, F. 1975. Distribution and ecology of skipjack tuna, Katsuwonus pelamis, in an offshore area of the eastern tropical Pacific Ocean. Fishery Bulletin 73:382-411.
Briggs, K.T., Tyler, W.B., Lewis, D.B. & Carlson, D.R. 1987. Bird communities at sea off California: 1975 to 1983. Studies in Avian Biology 11:1-74.
Brinton, E. 1967. Vertical migration and avoidance capability of Eupausiids in the California Current. Limnology and Oceanography 12:451-483.
Brown, R.G.B. 1980. Seabirds as marine animals. In: Burger, J., Olla, B.L., and Winn, H.E. (eds.). Behavior of marine animals. New York; Plenum Press:1-39.
Cairns D.K. & Schneider, D.C. 1990. Hot spots in cold water: feeding habitat selection by thick-billed murres. Studies in Avian Biology 14:52-60.
Diamond, A.W. 1975a. Biology and behaviour of frigatebirds Fregata spp. on Aldabra Atoll. Ibis 117:302-323.
Diamond, A.W. 1975b. The biology of tropicbirds at Aldabra Atoll, Indian Ocean. Auk 92:16-39.
Diamond, A.W. 1983. Feeding overlap in some tropical and temperate seabird communities. Studies in Avian Biology 8:24-46.
Duffy, D.C. 1983. The foraging ecology of Peruvian seabirds. Auk 100:800-810.
Erikstad, K.E., Moum, T. & Vader, W. 1990. Correlations between pelagic distribution of Common and Brünnich’s guillemots and their prey in the Barents Sea. Polar Research 8:77-87.
Feare, C.J. 1981. Breeding schedules and feeding strategies of Seychelles seabirds. Ostrich 52:179-185.
Flint, E.N. & Nagy, K.A. 1984. Flight energetics of free-living Sooty Terns. Auk 101:288-294.
Flint. E.N. 1991. Time and energy limits to the foraging radius of Sooty Terns Sterna fuscata. Ibis 133:43-46.
Gaston, A.J., Collins, B.T. & Diamond, A.W. 1987. Estimating densities of birds at sea and the proportion in flight from counts made on transects of indefinite width. Canadian Wildlife Service Occasional Paper Number 59.
Götmark, F., Winkler, D.W. & Andersson, M. 1986. Flock-feeding on fish schools increases individual success in gulls. Nature 319:589-591.
Gould, P.J. 1967. Nocturnal feeding of Sterna fuscata and Puffinus pacificus. Condor 69:529.
Gould, P.J. 1971. Interactions of seabirds over the open ocean. Ph.D. Thesis. University of Arizona, Tucson, U.S.A.
Gould, P.J. & Forsell, D.J. 1989. Techniques for shipboard surveys of marine birds. United States Department of the Interior, Fish and Wildlife Service, Fish and Wildlife Technical Report 25.
Grebmeir, J.M. & Harrison, N.M. 1992. Seabird feeding on benthic amphipods facilitated by gray whale activity in the northern Bering Sea. Marine Ecology Progress Series 80:125-133.
Haney, J.C. 1985. Counting seabirds at sea from ships: comments on interstudy comparisons and methodological standardization. Auk 102:897-898.
Haney, J.C., Fristrup, K.M. & Lee, D.S. 1992. Geometry of visual recruitment by seabirds to ephemeral foraging flocks. Ornis Scandinavica 23:49-62.
Harrington, B.A., Schreiber, R.W. & Woolfenden, G.E. 1972. The distribution of the male and female Magnificent Frigatebirds, Fregata magnificens, along the gulf coast of Florida. American Birds 26:927-931.
Harrington, B.A. 1977. Winter distribution of juvenile and older Red-footed Boobies from the Hawaiian Islands. Condor 79:87-90.
Harrison, C.S., Hida, T.S. & Seki, M.P. 1983. Hawaiian seabird feeding ecology. Wildlife Monographs 85:1-71.
Harrison, C.S. & Seki, M.P. 1987. Trophic relationships among tropical seabirds at the Hawaiian Islands. In: Croxall, J.P. (ed.). Seabirds. Feeding ecology and role in marine ecosystems. Cambridge; Cambridge University Press:301-326.
Heinemann, D., Hunt, G.L. Jr. & Everson, I. 1989. Relationships between the distributions of marine avian predators and their prey, Euphausia superba, in Bransfield Strait and southern Drake Passage, Antarctica. Marine Ecology Progress Series 58:3-16.
Hoffman, W., Heinemann, D. & Wiens, J.A. 1981. The ecology of seabird feeding flocks in Alaska. Auk 98:437-456.
Hulsman, K. 1980. Feeding and breeding strategies of sympatric terns on tropical islands. Proceedings of the International Ornithological Congress 17:984-988.
Hunt, G.L., Jr. & Schneider, D.C. 1987. Scale-dependent processes in the physical and biological environment of marine birds. In: Croxall, J.P. (ed.). Seabirds: feeding ecology and role in marine ecosystems. Cambridge; Cambridge University Press:7-41.
Hunt, G.L., Jr. 1988. The distribution of seabirds at sea: physical and biological aspects of their marine environment. Proceedings of the International 100 EO-G Meeting, Current Topics in Avian Biology, Bonn:167-171.
Hunt, G.L., Jr. 1990. The pelagic distribution of marine birds in a heterogeneous environment. Polar Research 8:43-54.
Hunt, G.L., Jr., Harrison, N.M. & Cooney, R.T. 1990. The influence of hydrographic structure and prey abundance on foraging of Least Auklets. Studies in Avian Biology 14:7-22.
Hunt, G.L., Jr. 1991. Marine ecology of seabirds in polar oceans. American Zoologist 31:131-142.
Hunt, G.L., Jr., Heinemann, D. & Everson, I. 1992. Distributions and predator-prey interactions of Macaroni Penguins, Antarctic Fur Seals, and Antarctic Krill near Bird Island, South Georgia. Marine Ecology Progress Series 86:15-30.
Hunt, G.L., Jr., Coyle, K.O., Hoffman, S., Decker, M.B. & Flint, E.N. 1996. Foraging ecology of Short-tailed Shearwaters near the Pribilof Islands, Bering Sea. Marine Ecology Progress Series 141:1-11.
Imber, M. J., Cruz, J.B., Grove, J.S., Lavenberg, R.J., Swift, C.C. & Cruz, F. 1992. Feeding ecology of the Dark-rumped Petrel in the Galápagos Islands. Condor 94:437-447.
King, W.B. 1970. The trade wind zone oceanography pilot study part VII: observations of sea birds March 1964 to June 1965. U.S. Fish and Wildlife Service Special Scientific Report. Fisheries No. 586.
Logerwell, E.A. & Hargreaves, N.B. 1996. The distribution of sea birds relative to their fish prey off Vancouver Island: opposing results at large and small spatial scales. Fisheries Oceanography 5:163-175.
Longhurst, A.R. & Pauly, D. 1987. Ecology of tropical oceans. San Diego; Academic Press.
Mörzer Bruyns, W.F.J. & Voous, K.H. 1965. Night-feeding by Sooty Tern. Ardea 53:79.
Murphy, G.I. & Ikehara, I.I. 1955. A summary of sightings of fish schools and bird flocks and of trolling in the central Pacific. Washington, D.C. U.S. Fish and Wildlife Special Scientific Report, Fisheries 154.
Obst, B.S. 1985. Densities of Antarctic seabirds at sea and the presence of the krill Euphausia superba. Auk 102:540-549.
Obst, B.S., & Hunt, G.L. Jr. 1990. Marine birds feed at gray whale mud plumes in the Bering Sea. Auk 107:678-688.
Obst, B.S., Russell, R.W., Hunt, G.L. Jr., Eppley, Z.A., & Harrison, N.M. 1995. Foraging radii and energetics of Least Auklets (Aethia pusilla) breeding on three Bering Sea islands. Physiological Zoology 68:647-672.
Olson, R.J. & Boggs, C.. 1986. Apex predation by Yellowfin Tuna (Thunnus albacares): independent estimates from gastric evacuation and stomach contents, bioenergetics, and cesium concentrations. Canadian Journal of Fisheries and Aquatic Sciences 43:1760-1775.
Peters, R.H. 1983. The ecological implications of body size. Cambridge; Cambridge University Press.
Philbrick, V.A., Fiedler, P.C., Reilly, S.B., Pitman, R.L., Ballance, L.T., Thomas, G.G. & Behringer, D.W. 1991. Report of ecosystem studies conducted during the 1990 eastern tropical Pacific dolphin survey on the research vessel David Starr Jordan. NOAA Technical Memorandum, NOAA-TM-NMFS-SWFC-160.
Piatt, J.F. 1990. The aggregative response of Common Murres and Atlantic Puffins to schools of capelin. Studies in Avian Biology 14:36-51.
Pitman, R.L. 1986. Atlas of seabird distribution and relative abundance in the eastern tropical Pacific. Administrative Report LJ-86-02C. NMFS, Southwest Fisheries Center, P.O. Box 271, La Jolla, CA 92038.
Pitman, R.L. & Ballance, L.T. 1990. Daytime feeding by Leach’s Storm-Petrel on a midwater fish in the eastern tropical Pacific. Condor 92:524-527.
Pitman, R.L. & Ballance, L.T. 1992. Parkinson’s Petrel distribution and foraging ecology in the eastern Pacific: aspects of an exclusive feeding relationship with dolphins. Condor 94:825-835.
Pocklington, R. 1979. An oceanographic interpretation of seabird distribution in the Indian Ocean. Marine Biology 51:9-21.
Ribic, C.A. & Ainley, D.G. 1997. The relationships of seabird assemblages to physical habitat features in Pacific equatorial waters during spring 1984-1991. ICES Journal of Marine Science 54:593-599.
Ribic, C.A., Davis, R., Hess, N. & Peake, D. 1997. Distribution of seabirds in the northern Gulf of Mexico in relation to mesoscale features: initial observations. ICES Journal of Marine Science 54:545-551.
Schmidt-Nielsen, K. 1984. Scaling. Why is animal size so important? Cambridge; Cambridge University Press.
Schneider, D.C. & Piatt, J.F. 1986. Scale-dependent correlation of seabirds with schooling fish in a coastal ecosystem. Marine Ecology Progress Series 32:237-246.
Schneider, D.C., Pierotti, R. & Threlfall, W. 1990. Alcid patchiness and flight direction near a colony in eastern Newfoundland. Studies in Avian Biology 14:23-35.
Schneider, D.C. 1991. The role of fluid dynamics in the ecology of marine birds. Oceanography and Marine Biology Annual Review 29:487-521.
Schneider, D.C. 1994. Quantitative ecology. Spatial and temporal scaling. San Diego; Academic Press:395 pp.
Schreiber, R.W. & Hensley, D.A. 1976. The diets of Sula dactylatra, Sula sula, and Fregata minor on Christmas Island, Pacific Ocean. Pacific Science 30:241-248.
Schreiber, R.W. & Chovan, J.L. 1986. Roosting by pelagic seabirds: energetic, populational, and social considerations. Condor 88:487-492.
SeaWiFS Project Image Archive. Global Sea Surface Temperature Maps [online]. Cited 21 July 1998. Available from World Wide Web: http://psbsgi1.nesdis.noaa.gov:8080/PSB/EPS/SST/contour.html.
SeaWiFS Project Image Archive. Nimbus-7 Coastal Zone Color Scanner [online]. Cited 21 July 1998. Available from World Wide Web: http://seawifs.gsfc.nasa.gov/SEAWIFS/IMAGES/GALLERY.html.
Serventy, D.L., Serventy, V. & Warham, J. 1971. The handbook of Australian seabirds. Sydney; A.W. and A.H. Reed.
Shealer, D.A. 1996. Foraging habitat use and profitability in tropical Roseate Terns and Sandwich Terns. Auk 113:209-217.
Spear, L.B., Nur, N. & Ainley, D.G. 1992. Estimating absolute densities of flying seabirds using analyses of relative movement. Auk 109:385-389.
Spear, L.B. & Ainley, D.G. 1997a. Flight behaviour of seabirds in relation to wind direction and wing morphology. Ibis 139:221-233.
Spear, L.B. & Ainley, D.G. 1997b. Flight speed of seabirds in relation to wind speed and direction. Ibis 139:234-251.
Steadman, D.W. 1995. Prehistoric extinctions of Pacific island birds: biodiversity meets zooarchaeology. Science 267:1123-1131.
Steadman, D.W. 1997. Extinctions of Polynesian birds: reciprocal impacts of birds and people. In: Kirch, P.V., and Hunt, T.L. (eds.). Historical ecology in the Pacific islands. New Haven; Yale University Press:51-79.
Tasker, M.L., Jones, P.H., Dixon, T. & Blake, B.F. 1984. Counting seabirds at sea from ships: a review of methods employed and a suggestion for a standardized approach. Auk 101:567-577.
Van Franeker, J.A. 1990. Methods for counting seabirds at sea: a plea for comparative research. Sula 4:85-89.
Veit, R.R. & Hunt, G.L. Jr. 1991. Broadscale density and aggregation of pelagic birds from a circumnavigational survey of the Antarctic Ocean. Auk 108:790-800.
Veit, R.R., Silverman, E.D. & Everson, I. 1993. Aggregation patterns of pelagic predators and their principal prey, Antarctic Krill, near South Georgia. Journal of Animal Ecology 62:551-564.
Warham, J. 1977. Wing loadings, wing shapes, and flight capabilities of Procellariiformes. New Zealand Journal of Zoology 4:73-83.
Weimerskirch, H. & Wilson, R.P. 1992. When do Wandering Albatrosses Diomedea exulans forage? Marine Ecology Progress Series 86:297-300.
Wiens, J.A. 1989. Spatial scaling in ecology. Functional Ecology 3:385-397.
Fig. 1. Global sea surface temperature. The geographical extent of tropical oceans, defined as water ³ 23°C (Ashmole 1971), can clearly be seen. (Modified from SeaWiFS Project Image Archive. Global Sea Surface Temperature Maps [online]. Cited 21 July 1998.)

Fig. 2. Global sea surface chlorophyll concentration. Note that productivity is lowest in the tropics and central gyres. (Modified from SeaWiFS Project Image Archive. Nimbus-7 Coastal Zone Color Scanner [online]. Cited 21 July 1998.)